| |  | |
Most of the site will reflect the ongoing surgical activity of Prof. Munir Elias MD., PhD. with brief slides and weekly activity. For reference to the academic and theoretical part, you are welcome to visit neurosurgery.tv |
|
Functional Neurosurgery
functionalneuro.surgery
Functionalneurosurgery.net
IOM Sites
iomonitoring.org
operativemonitoring.com
Neurosurgical Sites
neurosurgery.art
neurosurgery.me
neurosurgery.mx
skullbase.surgery
Neurosurgical Encyclopedia
neurosurgicalencyclopedia.org
Neurooncological Sites
acousticschwannoma.com
craniopharyngiomas.com
ependymomas.com
gliomas.info
gliomas.uk
meningiomas.org
neurooncology.me
pinealomas.com
pituitaryadenomas.com
Neuroanatomical Sites
humanneuroanatomy.com
microneuroanatomy.com
Neuroanesthesia Sites
neuro-anesthessia.org
Neurobiological Sites
humanneurobiology.com
Neurohistopathological
neurorhistopathology.com
Neuro ICU Site
neuroicu.info
Neuroophthalmological
neuroophthalmology.org
Neurophysiological Sites
humanneurophysiology.com
Neuroradiological Sites
neuroradiology.today
NeuroSience Sites
neuro.science
Neurovascular Sites
vascularneurosurgery.com
Personal Sites
cns.clinic
Spine Surgery Sites
spine.surgery
spondylolisthesis.info
paraplegia.today
Stem Cell Therapy Site
neurostemcell.com

Inomed Stockert Neuro N50. A versatile
RF lesion generator and stimulator for
countless applications and many uses

Multigen RF lesion generator .
|
 |

| | |
 |
INDUSTRY INDUCED
COMPLICATIONS
|
 |
All the time the surgeon is responsible for complications.
This traditional view to the events must be changed. Here is an example:
|
|
This coated visorb manufactured by CP Medical, Portland , OR USA, purchased the
2 months by Shmaisani hospital was given to me and I performed 11 surgeries with this
material. Among patients was a
doctor and another one is the father of the doctor. This material caused disastrous events to all my eleven patients. The usual
clinical picture starts not immediately, but the third or fourth
postoperative day, when a subcutaneous massive serous fluid start to exudate in massive quantity, that you start to think that CSF leak took place. The
father of the doctor to whom the surgery was undertaken for OPLL was kept in hospital for three weeks with the thought that CSK leak is coming out and he was covered with vancomycin and mannitol. The patient was kept in a position to decrease the flow of the proposed CSF leak. During that time the wound was open, but the subcuticular material was in place. Accidentally , I removed this material and within three days all the reactions subsided and the patient was discharged. You can see the operative video record. You can also see the video record of
patient with LCS, how required debridement and suturing by nylon, after what the wound healed normal. Even the doctor with 2 cm incision for PLD still complaining of dehiscence and mostly she will require debridement under G.A for this 2 cm length incision. The company has no address to contact with and the dealers with such materials are a loose number to take the responsibility for such events. In case if some authorized personality follow this event , this material must be immediately withdrawn from the market. Thanks God that the series of patients were understandable and cooperative. But thousands of JDs were spent to correct these sequelae.
Later the FDA was involved with this problem and complete report
was given and they promised to resolve the problem.
The material issued in
08-April-2005 |
|
Case presentation:
|
|
|
A young 25 years old patient was operated 2 months ago, after
what he got persistent CSF leak. The patient was seen 05-11-2004
with mild meningism, lethargic, without fever and no
considerable neurologic deficit. The patient performed MRI with
contrast 05-11-2004. It showed a huge defect at the right
cribriform plate with air filling the entire ventricular
system with massive dilatation of the right lateral ventricle,
which was reaching the base of anterior fossa.
The patient was operated
06-November-2004: Through subfrontal approach
right side, repair of the dural defect and sealing of the base
by muscle and bone graft was performed. Video available. It is
very rare to see such complication, that reaching the
ventricular system, making wide communication between the
ventricular system and the sinuses. |
1.Intraoperative
complications not related with the surgery.
1.
Intraoperative CVA of the left cerebral
hemisphere during discectomy for PCD C4-5 and C5-6 with fusion.
2.Postsurgery not
related complications/events:
1. Acute necrotizing pancreatitis
2. Massive pulmonary embolism.
Case 17-January-2008
3. Drug Induced Complications: There is a material of five patients with unknown to
me complications 4 of them fatal and one survived after laparatomy.
It was impossible to know what is going on, until the last case gave
the clue for the reason of these complications. The common
dominator between all these case, that all of them during the last 2
months, that they received dexametasone 16 mg three times a
day for minimum 2 days. After analyzing the cause, it became clear,
that Decadron was replaced by dexametasone of Egyptian brand (
EIPICO); which was used the last 2 months in the hospital, where my
activities are going on.
Here the list of patients in retrospective
retrograde chronological order: 1.
Medulloblastoma in young 36 year woman.
2. A young chap with dorsal neurenteric
cyst with massive GI bleeding requiring laparatomy.
3. A case after decompressive cervical
coporectomy complicated by acute necrotizing pancreatitis.
4. Recurrent brain-stem infarction with
edema after successful endarterectomy.
5. Recurrent craniopharyngioma with huge
intraventricular extension. During the 26 year
experience, I have the ability to predict the possible complications
in certain pathologies, but with introduction of various medications
from new companies with unknown standards, the possibilities of
complications is becoming wide and the prediction for complication
is loosing the clear picture. Thanks to God that I work in one
place, and I have the ability to retrospectively catch the problem,
but may be there are thousands of patient paying their life for
corrupted medical companies. This part issued
14-August-2006
Contrast agents used in MRI investigations.
 |
A
patient performed more
than 10 times over the years MRI investigations
using contrast media without complications. The
center was using Magnovist. At this time he was
given MultiHans and this could cost to him his
life due to rapid allergic anaphylactic shock
which was treated immediately. |
 |
Another known complication
could have place is
nephrogenic systemic fibrosis.
|
|
 |
|
2. Surgical products causing complications
during surgery:
1: BloodSTOP ix:
This product was offered for me to use it in critically vascular tumors to
decrease bleeding, but in the contrary, it caused many problems and in the
end result it was the source for further bleeding. For more details please
refer to the presentation case.
25-December-2009.
2.
Tsunamimedical vertebroplasty bone cement slipping to the
intraossal veins of the near by vertebrae and the epidural veins causing
vascular compromise to the roots. For demonstration,
Click here|
3.
Cefazolin
causing hypocoagulation with delayed oozing of the clean surgical wound,
necessitating delayed discharge of the patients. After revision of the
directories of infection control, we were advised to start and continue
Cefazolin I/V Jordanian production, to prevent infection in clean
neurosurgical patients. All the patients showed the same picture of
continuous oozing of the wound and delayed discharge of the patients from
the hospital. After stopping the product, the wounds were clean and we could
discharge the patients. The cases were during July 2014:
Case one,
case two, and
case three. This item was added
07-July-2014.
3.Postsurgery
related complications/events:
1. CSF pocket after removal of
right S3 Tarlov cyst.
2. Immediate postoperative CSF leak.
3. PEEK Satellite Nucleus Replacement Sphere
with discitis.
4. Hydr'Os bone graft injected to the
intradiscal space, which gradually dissipated causing inflammatory reactions
with events mimicking swollen root, then ganglion of the lateral facet and
triggering recurrence of huge extrusion.
5. Postoperative tracheostomy
related complication.
4.Bad surgeon related complications:
1. Unstable cervical spine after
supposed to be discectomy of C6-7.
So as to have digital data registering the complications in
evolution and escalation, we introduced the best in the world
ISIS Highline Inomed IOM. It started
30-August-2007 and became in common use for all surgeries since that time.
4. MRI Induced complications:
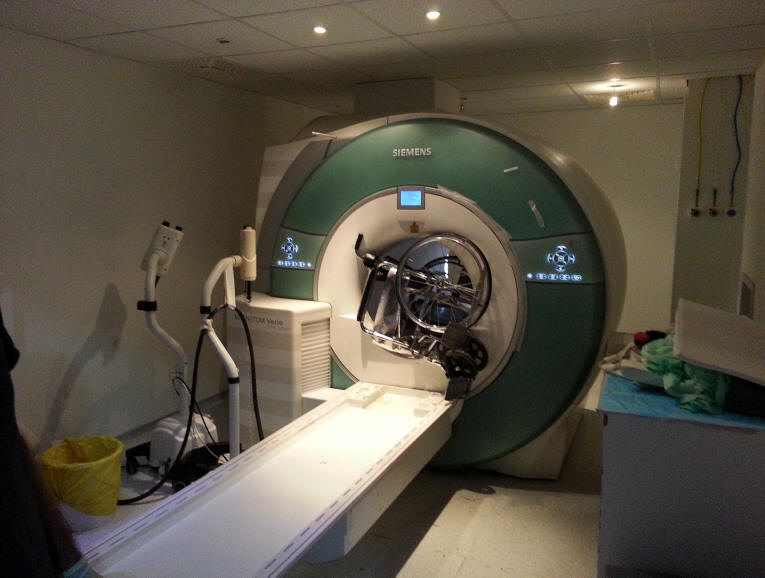


Figure: 1,2,3 showing the wheelchair stuck to the MRI from deferent
angles. This is the result of negligence of the technical staff and
disruption of the regulations, allowing non certified personnel entering the
dangerous areas. This happened in facility in one of the respected hospitals
in Amman, Jordan 12-July-2014.
5.
Medical Error Is Third Leading Cause of Death After Heart Disease and Cancer
in USA.
Published in Medscape Neurology 04-May-2016.
|
 |
|